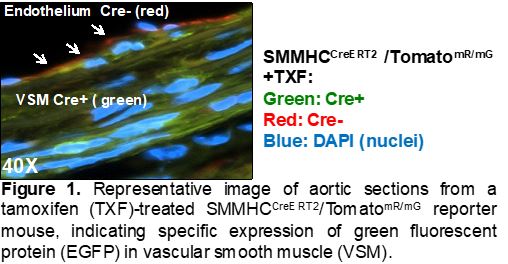
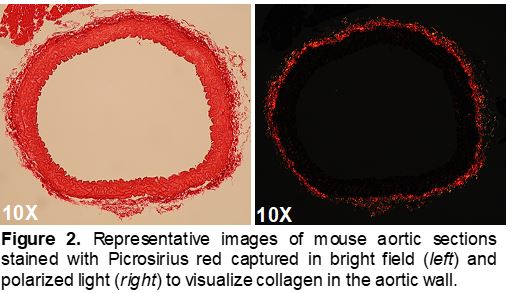
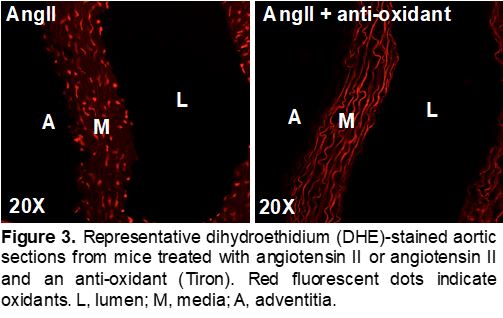
Seta Lab
Dr. Seta’s laboratory studies the basic mechanisms of vascular diseases, with an emphasis on the biology of the vascular smooth muscle. Our group seeks to understand the molecular mechanisms of arterial stiffening and aortic aneurysms/dissections, two degenerative vascular conditions for which there are no therapies. Hardening of elastic arteries is an age-related maladaptive remodeling which occurs independently of atherosclerosis but is comparably harmful to the cardiovascular system.
We were the first to show that arterial stiffness develops rapidly in a model of diet-induced obesity/metabolic syndrome and precedes the development of hypertension. In collaboration with investigators of the Framingham Heart Study, based at Boston University, we identified a genetic locus with genome-wide association to increased arterial stiffness. This “arterial stiffness” locus functions as enhancer for the the zinc finger protein Bcl11b. A specific project currently underway is the basic investigation of the role of Bcl11b in the regulation of vascular smooth muscle contractile phenotype and arterial functional integrity and how they causally relate to the development of arterial stiffening.
We also identified the lysine deacetylase sirtuin-1, known as the “longevity” gene and a molecular target of caloric restriction, as a crucial regulator of vascular homeostasis. We demonstrated that sirtuin-1 genetic overexpression or pharmacological activation exerts beneficial anti-oxidant and anti-inflammatory effects in the aortic wall, protecting against obesity-induced arterial stiffness and cardiac diastolic dysfunction. On the opposite, lack of sirtuin-1 in vascular smooth muscle caused aortic dissection in mice treated with angiotensin II. Current studies are examining the therapeutic potential of sirtuin-1 against aortic aneurysms in a mouse model of Marfan’s syndrome, a condition characterized by aortic enlargements at risk of rupture or dissection. Our laboratory employs state-of-the art in vivo, ex vivo and in vitro approaches, including high-resolution ultrasound echocardiography, radiotelemetry, high-fidelity pressure catheters and pressurized myography, to assess pulse wave velocity, the gold standard measure of arterial stiffness; active and passive properties of blood vessels; imaging and characterization of aortic aneurysms. Our goal is to identify molecular targets to develop novel translational therapies that can prevent and treat dreadful vascular diseases.
Lab members
 |
Francesca Seta, PhD |
Principal Investigator Associate Professor |
 |
Enkhajal Budbazar | Research Scientist |