 The work of Avrum Spira and his group may eventually lead to a simple screening for lung cancer. Photo by Cydney Scott
The work of Avrum Spira and his group may eventually lead to a simple screening for lung cancer. Photo by Cydney Scott
Lung cancer is the deadliest form of cancer in the United States—and in the world. According to the National Cancer Institute, it accounts for more than a quarter of cancer deaths in this country, killing about 158,000 people in 2016.
Because lung cancer is so lethal, many current and former smokers undergo precautionary CT scans of the chest. These scans can detect small lesions in the lungs that may be an early sign of cancer, but false positives can lead to invasive and unnecessary lung biopsies. New research points to a better path to diagnosis: a genomic test that may eventually require only a simple nasal swab. The findings appeared online in the Journal of the National Cancer Institute (JNCI) on February 27, 2017.
“The idea that we can sample cells from the nose and tell if someone has lung cancer—that is sort of out there,” says JNCI paper senior author Marc Lenburg, a School of Medicine professor of medicine. “To sample cells that far from the tumor and still find a genetic signal is really exciting.”
The findings build on previous research led by Lenburg and JNCI paper corresponding author Avrum Spira (ENG’02), Alexander Graham Bell Professor in Health Care Entrepreneurship and a MED professor of medicine, pathology, and laboratory medicine. In 2015, the researchers reported in the New England Journal of Medicine that a genomic test they’d developed could detect lung cancer in a section of the bronchial airway far removed from the tumor. The test, now called Percepta, was acquired by the genomic diagnostics company Veracyte, released that year, and received Medicare approval in late 2016.
During the Percepta test, which is performed at the same time as a bronchoscopy, a doctor uses a small brush to collect cells in the upper bronchial airway. Spira discovered that these cells, while appearing normal, reside in the cancer’s “field of injury” and contain genetic markers that signal a high likelihood of cancer elsewhere in the lung.
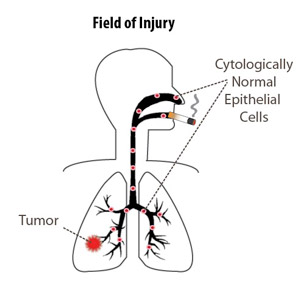 Lung cancer’s “field of injury” extends well beyond the tumor. New work by Avrum Spira and colleagues demonstrates genetic changes in nasal cells of patients who went on to develop lung cancer. Courtesy of Avrum Spiranet
Lung cancer’s “field of injury” extends well beyond the tumor. New work by Avrum Spira and colleagues demonstrates genetic changes in nasal cells of patients who went on to develop lung cancer. Courtesy of Avrum Spiranet
The problem, says Spira, is that “there is a subset of people with pulmonary lesions who do not undergo a bronchoscopy as part of their diagnostic workup,” either because the suspicious lesions are very deep in the lung and inaccessible to the bronchoscope or because the patient suffers from other conditions—like heart failure or emphysema—that make a bronchoscopy difficult or dangerous. But if people with lung cancer have genomic alterations in the cells in the nose too, a nasal swab could potentially serve the same purpose as a bronchial brush.
To explore this possibility, Spira and Lenburg analyzed genomic changes in nasal swabs from patients recruited for the Percepta clinical trials. “We had previously collected the nasal samples,” says Spira. “But we thought that genomic changes in the nose that associate with lung cancer would be more difficult to detect than those found in the bronchial airway.” This proved to be true, but recent improvements in computational approaches to genomic data analysis allowed the team to find a lung cancer signal in the nasal samples. They found 535 genes that were expressed differently in the nasal passages of patients who went on to develop lung cancer, but not in patients with benign lesions. Of those genes, 30 were sufficiently distinctive to be useful as a diagnostic biomarker.
The researchers say that the nasal test needs additional testing. “The people in the original study were already at substantially elevated risk for lung cancer,” says Lenburg. “We need to confirm that these results are valid for a wider population.” Nevertheless, the researchers hope that their findings will eventually lead to a widely used test.
“In the short term, we’ll use the nasal biomarker as a diagnostic test for people who have something suspicious on a CT scan of the chest, but are not undergoing bronchoscopy,” says Spira. “But eventually, the nasal test could impact a lot more people as a mass screening tool for lung cancer. That’s the really exciting potential.”