Maria V. Panchenko, Ph.D.
Assistant Professor
Contact
Boston University School of Medicine
Department of Pathology & Laboratory Medicine
670 Albany Street, #416
Boston, MA 02118
.
.
Education
- BA/MS in Biochemistry, Moscow State University
- Ph.D in Biochemistry, Moscow State University, Department of Biochemistry
Training/Positions
- Postdoctoral Fellow, Department of Biochemistry, Boston University School of Medicine, Boston, MA
- Instructor of Medicine, Nephrology, Department of Medicine, Boston University Medical Center, Boston, MA
Contact
Boston University
School of Medicine
Department of Pathology & Laboratory Medicine
670 Albany Street, #416
Boston, MA 02118
mpanch@bu.edu
Research Interests:
Our lab is interested in the area of epithelial cell biology and homeostasis in mammals. A single layer of epithelial cells lining tubules are found in several organs and serve protective and active functions. Thus, the differentiated epithelial cells are known to mediate a number of functions, including transmembrane transport, filtration and secretion in and out of the lumen. An interesting feature of epithelial cells is that they are able to de-differentiate and enter the cell cycle which is important for the maintenance of the lining, or proliferate after an injury to replenish for the lost cells and aid in organ regeneration (kidney tubular epithelia). Epithelial cell progenitors and stem cells such as those found in the bottom of colon crypts, divide to provide continuous renewal of differentiated columnar epithelial lining. The rates of epithelial cells turnover varies and depends on their specific function in a given organ. The regulation of the cell cycle is crucial for the homeostasis and regeneration of epithelia in vivo.
Accurate replication, segregation, and orientation of chromatin ensure genetic integrity and represent most important events during the epithelial cell division. Errors may result in either insufficient regeneration (degenerative pathology) or excessive uncontrolled proliferation (cancer). We are specifically interested in factors that sense and directly link chromatin integrity with the cell division, which is an exciting area of epithelial cell biology and is highly relevant to human diseases.
We use in vitro cell culture models, transgenic mice models, and human specimens to discover and validate factors that interact with chromatin and control cell cycle progression.
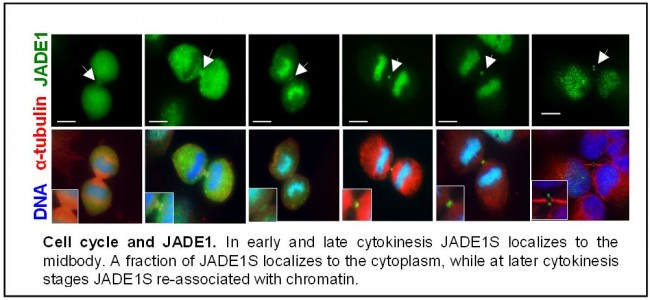
Project 1. Previously we and others identified the PHD zinc finger family JADE1/2/3 proteins as members of at least two HAT complexes (HBO1 and TIP60). We reported that the function of JADE1 is to target a HAT to chromatin via JADE1 PHD zinc fingers and enable acetylation of bulk histone H4. Acetylation and de-acetylation of specific lysine residues within the N-terminus of histone H4 is thought to be one of the key events required for chromatin replication and homeostasis during the cell cycle. In addition, control of chromatin modification is required for proper segregation in mitosis and cytokinesis. Recently we discovered unexpected role of JADE1, which defined JADE1 as novel cytokinesis checkpoint factor controlling the cell cycle progression. We are examining JADE1 pathway in epithelial cell proliferation. Project 1 is to identify the specific role in chromatin and cell division, mechanisms and molecular partners of JADE proteins.
For in vivo studies we choose kidney tubular and colon lining epithelia. These models are well characterized and are most suitable for the validation and translation of our molecular mechanistic findings. We use several models of acute and chronic kidney injury (AKI and CKI) and regeneration in mice which closely resembles human AKI and CKI – conditions known to associate with chronic and end stage kidney disease. We analyze colons of humans and mice which are ideal models to investigate molecular events during epithelial proliferation, differentiation, and apoptosis. We use transgenic mice models to determine the in vivo outcomes of gene depletion or overexpression.
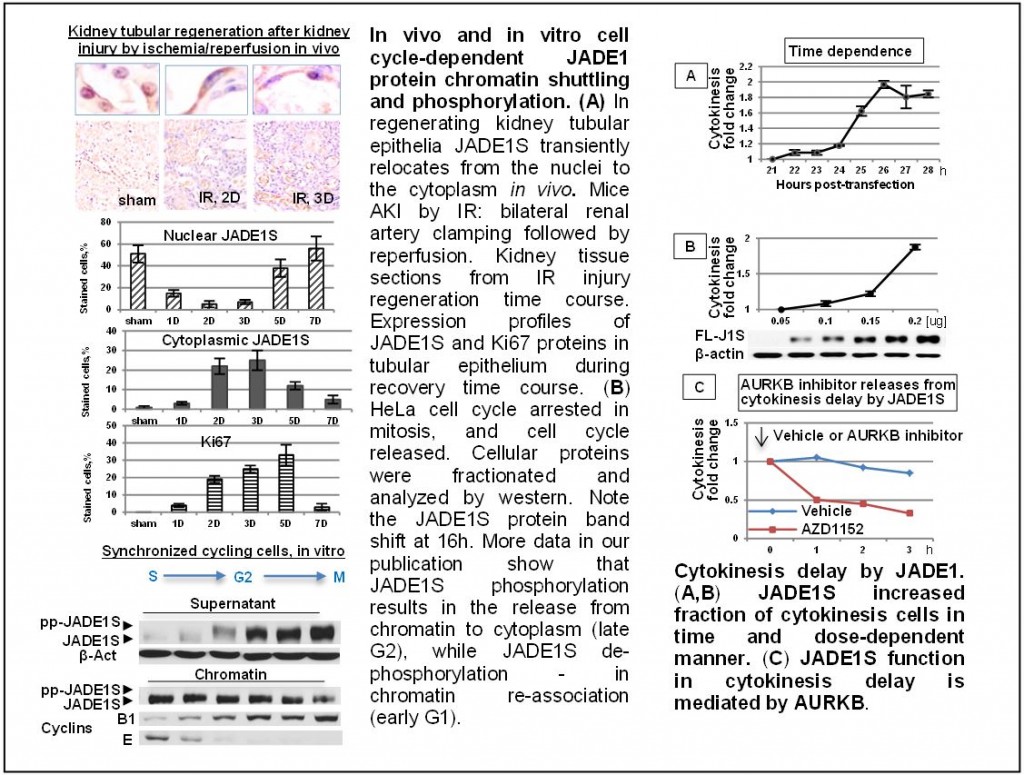
Project 2. Kidney tubular injury and regeneration. This project examines biological role of the JADE1-HBO1 complex in kidney epithelial cell cycle. We used mice model of acute kidney injury after ischemia-reperfusion and recently reported the activation of JADE1-HBO1 complex during tubular cells regeneration. By using complementary in vitro and in vivo transgenic mice models, we are dissecting mechanisms by which HBO1-JADE1 participate in tissue regeneration. Our long term goal is to find out how these complexes regulate cell cycle in regenerating normal tissues, as well as in diseases with abnormal cell proliferation, including cancer.
Project 3. Cell cycle control and colon cancer. Several HAT complexes have been linked to diseases, including cancers. Moreover, PHD zinc fingers of known HATs and other proteins such as p300, MOZ, MORF, AIRE, INGs are targets of mutations in several diseases. JADE1 and HBO1 may play a role in pathogenesis of colon adenocarcinomas.
Lab Members
Bo Shao, M.S. Northeastern University, Research Technician
Justin Cho, NIDDK STEP-UP Fellow (year 2016), (currently B.A. Student at Williams College, MA)
Nirodhini Siriwardana, Ph.D., Research Associate Scientist (currently Senior Scientist, Merck)
Rosana Meyer, M.D., Senior Lab Manager (currently postdoctoral in N.Rahimi lab, BUSM)
Charlie Aoun, B. Sc. Student, BU (currently Clinical Research Coordinator, UCSF)
Konstantine Spirin, Ph.D., Research Associate Scientist
Somdutta Mitra, M.S. Research Technician (currently Senior Research Associate at FORMA Therapeutics, Inc.)
Joseph Haegele, B. Sc. Research Technician (currently Ph.D. Student at Cornell University)
Sherry Blackmon, B. Sc. Research Technician (currently DVM/PhD Student at Mississippi State University)
Alice Lee, 2012 Summer UROP award and Fellowship, B. Sc. Candidate
Funding
1R01DK087910
NIH NIDDK
“Histone Acetyl Transferase HBO1 in kidney epithelial cell cycle”
Role: PI This 5 year project investigates role of JADE1-HBO1 in the cell cycle in kidney epithelial cell cycle and regeneration after hypoxic injury.
Positions Available:
Graduate (Ph.D. or M.S.) Student
Selected Publications
Nolin A, Mulhern R, Panchenko M, Pisarek-Horowitz A, Wang Z, Shirihai O, Borkan S, and Havasi A. 2016. Proteinuria causes dysfunctional autophagy in the proximal tubule. American Journal of Physiology (accepted)
Panchenko MV. 2016. Structure, function and regulation of Jade Family PHD Finger 1 (JADE1) GENE 589(1):1-11 PMID: 27155521
Siriwardana, NS, Meyer RD, and Panchenko MV* (2015): The novel function of JADE1S protein in cytokinesis of epithelial cells. Cell Cycle 14:2821-2834 PMID: 26151225 * Comment in Cell Cycle by Prekeris R (2015): Cut or NoCut: the Role of JADE1S in Regulating Abscission Checkpoint Cell Cycle
Siriwardana NS, Meyer R, Havasi R, Dominguez I, and Panchenko MV* (2014): Cell cycle-dependent Chromatin shuttling of HBO1-JADE1 histone acetyl transferase (HAT) complex. Cell Cycle Jun 15;13(12):1885-901 PMID: 24739512 *Comment in Cell Cycle by Calvi BR (2014): HBO1:JADE1 at the cell cycle chromatin crossroads.
Havasi, A., J. A. Haegele, J. M. Gall, S. Blackmon, T. Ichimura, R. G. Bonegio, and M. V. Panchenko. Histone acetyl transferase (HAT) HBO1 and JADE1 in epithelial cell regeneration. Am J Pathol 152-162.
Foy, R. L., V. C. Chitalia, M. V. Panchenko, L. Zeng, D. Lopez, J. W. Lee, S. V. Rana, A. Boletta, F. Qian, L. Tsiokas, K. B. Piontek, G. G. Germino, M. I. Zhou, and H. T. Cohen. 2012. Polycystin-1 regulates the stability and ubiquitination of transcription factor Jade-1. Hum Mol Genet 5456-5471.
Foy, R. L., I. Y. Song, V. C. Chitalia, H. T. Cohen, N. Saksouk, C. Cayrou, C. Vaziri, J. Cote, and M. V. Panchenko. 2008. Role of Jade1 in the histone acetyltransferase (HAT) HBO1 complex. J Biol Chem 283:28817-28826.
Chitalia, V. C., R. L. Foy, M. M. Bachschmid, L. Zeng, M. V. Panchenko, M. I. Zhou, A. Bharti, D. C. Seldin, S. H. Lecker, I. Dominguez, and H. T. Cohen. 2008. Jade-1 inhibits Wnt signalling by ubiquitylating beta-catenin and mediates Wnt pathway inhibition by pVHL. Nat Cell Biol 1208-1216.
DiCamillo, S. J., S. Yang, M. V. Panchenko, P. A. Toselli, E. F. Naggar, C. B. Rich, P. J. Stone, M. A. Nugent, and M. P. Panchenko. 2006. Neutrophil elastase-initiated EGFR/MEK/ERK signaling counteracts stabilizing effect of autocrine TGF-beta on tropoelastin mRNA in lung fibroblasts. Am J Physiol Lung Cell Mol Physiol L232-243.
Zhou, M. I., R. L. Foy, V. C. Chitalia, J. Zhao, M. V. Panchenko, H. Wang, and H. T. Cohen. 2005. Jade-1, a candidate renal tumor suppressor that promotes apoptosis. Proc Natl Acad Sci U S A 11035-11040.
Panchenko, M. V., M. I. Zhou, and H. T. Cohen. 2004. von Hippel-Lindau partner Jade-1 is a transcriptional co-activator associated with histone acetyltransferase activity. J Biol Chem 56032-56041.
DiCamillo, S. J., I. Carreras, M. V. Panchenko, P. J. Stone, M. A. Nugent, J. A. Foster, and M. P. Panchenko. 2002. Elastase-released epidermal growth factor recruits epidermal growth factor receptor and extracellular signal-regulated kinases to down- regulate tropoelastin mRNA in lung fibroblasts. J Biol Chem 18938-18946.
Panchenko, M. V., H. W. Farber, and J. H. Korn. 2000. Induction of heme oxygenase-1 by hypoxia and free radicals in human dermal fibroblasts. Am J Physiol Cell Physiol C92-C101.
Panchenko, M. V., W. G. Stetler-Stevenson, O. V. Trubetskoy, S. N. Gacheru, and H. M. Kagan. 1996. Metalloproteinase activity secreted by fibrogenic cells in the processing of prolysyl oxidase. Potential role of procollagen C-proteinase. J Biol Chem 7113-7119.
Graven, K. K., R. F. Troxler, H. Kornfeld, M. V. Panchenko, and H. W. Farber. 1994. Regulation of endothelial cell glyceraldehyde-3-phosphate dehydrogenase expression by hypoxia. J Biol Chem 24446-24453.
Kagan, H. M., V. B. Reddy, M. V. Panchenko, N. Nagan, A. M. Boak, S. N. Gacheru, and K. M. Thomas. 1995. Expression of lysyl oxidase from cDNA constructs in mammalian cells: the propeptide region is not essential to the folding and secretion of the functional enzyme. Journal of Cellular Biochemistry 329-338.