The Lehman Lab
Research

Our overarching goal is to understand all aspects of thin filament regulation of cardiac muscle contractile function by characterizing the system at a fundamental structural level. Likewise, we aim to use our structural tools, including cryogenic electron microscopy and computational biochemistry protocols that we developed to define the root causes and potential severity of mutation based-thin filament-linked dysfunction that lead to cardiomyopathy.
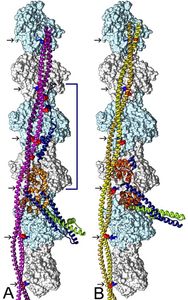
The long-held paradigm that troponin-regulated tropomyosin movement on actin of thin filaments controls myosin-head access to actin-binding sites and thus striated muscle activity was first formulated by Hanson, Lowy, and Huxley, and then supported by EM-reconstructions generated by my laboratory together with my colleagues Vibert and Craig. Recently, it has also become clear that regulated release and recruitment of myosin-heads from thick filaments and onto thin filaments augments tight control of muscle activity, increasing the need to formulate how myosin-head contact impacts thin filament troponin-tropomyosin dynamics.
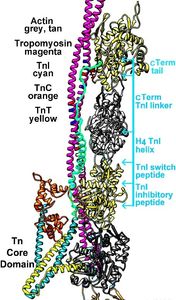
Ostensibly benign point mutations associated with cardiac muscle thin filament-linked proteins often cause subtle localized perturbation of filament structure that elicits significant hyper- or hypo-contractile behavior which over years can lead to cardiomyopathies. For example, many cases of hypertrophic (HCM), dilated (DCM), restrictive (RCM), and non-compaction (LVNC) cardiomyopathy are linked to point mutations in cardiac specific proteins that are our focus, viz. actin, myosin, troponin and tropomyosin. Indeed, our studies directly address regulation of cardiac muscle contractility at a fundamental molecular level where such disease-related “insults” may first originate and then lead to higher order dysfunction through diverse and still incompletely understood pathways. Given this complexity, it is essential to elucidate underlying structural changes and thus define early intervention strategies to prevent or delay disease progression, and later treat overt effects.
We take a “basic-science” approach while cognizant of the potential of our work to develop approaches to combat disease.
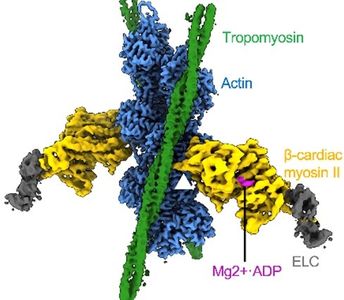
The rapidly accelerating progress in structural biology, driven by advances in cryo-EM and by the power of computational chemistry, has led to what is coined the “Resolution Revolution”; indeed, the cryo-EM of Yamada, Fujii and Namba confirmed the canonical steric model of thin filament regulation in remarkable detail. Most significantly, the possibility of precisely defining the dynamics of thin filament regulatory states at near-atomic scale is now accessible. Taking advantage of our expertise in cryo-EM and computational modeling, we published multiple papers that lay the foundation for our and colleagues future work. In particular, our studies highlight changing residue-to-residue contacts made by thin filament components with each other and myosin.
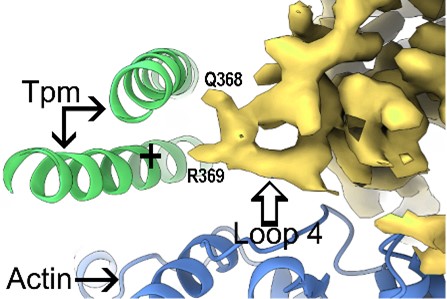
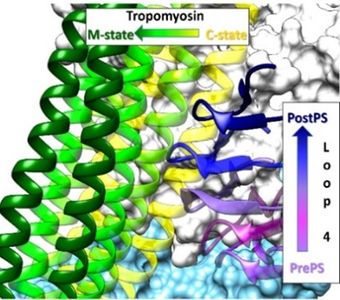
As well-positioned participants in the Resolution Revolution in structural biology, we take a lead in producing new paradigms revealing how cardiac thin filament regulation works at an atomic level and how disease-bearing mutants inexorably can lead to cardiomyopathy.
Recent Publications:
1. Chitre K, Karpicheva OE, King CJ, Rynkiewicz MJ, Fenwick AJ, Dawson JF, Foster DB, Lehman W, Cammarato A. (2026) Pseudo-acetylation of ACTC1 K326 and K328 promotes dysinhibition of reconstituted human cardiac thin filaments. J Mol Cell Cardiol. 212:10-15. doi: 10.1016/j.yjmcc.2025.12.008. Epub 2025 Dec 22. PMID: 41443503. “PMC Journal – In Process.”
2. Lehman W. (2026) The structural role of tropomyosin in regulating thin filament activation of actin-myosin interaction. Review. Subcell Biochem. 113:59-80. doi: 10.1007/978-3-032-05273-5_3. PMID: 41557229. “PMC Journal – In Process.”
3. Halder, S.S., Bellitto, J.G., Rynkiewicz, M.J., Cheung, F., Liu, D., Firlar, I., Bongiorno, A., Barry, M.E., Sewanan, L. Moore, J.R., Lehman, W., Campbell, S.G. (2025) Evaluating pathogenicity of TPM1 variants of unknown significance using in-silico and in-vitro models. J. Prec. Med. Health Dis. 2:100008. doi:10.1016/j.premed. “PMC Journal – In Process.”
4. Ramachandran, B., Rynkiewicz, M., Lehman, W. (2025) Velcro-binding by cardiac troponin-I traps tropomyosin on actin in a low-energy relaxed state. Biochem. Biophys. Res. Commun. 757:151595 doi: 10.1016/j.bbrc.2025.151595. PMCID: PMC11938286.
5. Wen, J., Campbell, S., Moore, J., Lehman, W., Rynkiewicz, M. (2025) Screening single nucleotide changes to tropomyosin to identify novel cardiomyopathy mutants. J. Mol. Cell Cardiol. 203:82-90. doi: 10.1016/j.yjmcc.2025.04.009. PMCID: PMC12135046.
6. Barry, M.E., Rynkiewicz, M.J., Wen, J., Tu, A.-Y., Regnier, M., Lehman, W., Moore, J.R. (2025) Dual role of cardiomyopathy tropomyosin-R160 in thin filament regulation: Insights into phosphorylation-dependent cardiac relaxation and mechanisms. Arch. Biochem. Biophys. 768:110380. doi:10.1016/j.abb.2025.110380. PMCID: PMC12276941.
7. Doran, M.H., Rynkiewicz, M.J., Despond,, E., Viswanathan, M.C., Madan, A. Chitre, K., Fenwick, A.J., Sousa, D., Lehman, W., Dawson, J.F., Cammarato, A. (2025) HCM-causing A331P actin enhances basal contractile activity and elicits resting muscle dysfunction. iScience 28(2):111816. doi: 10.1016/j.isci.2025.111816. PMC11841076.
8. Creso, J.G., Gokhan, I., Rynkiewicz, M., Lehman, W., Campbell, S., Moore, J.R. (2024) In silico and in vitro models reveal the molecular mechanisms of hypocontractility caused by TPM1 M8R. Frontiers Physio. 15 doi: 10.3389/fphys.2024.1452509. PMCID: PMC11392859.
9. Halder, S., Rynkiewicz, M.J., Kim, L., Barry, M.E., Zied, A.G.A., Sewanan, L.R., Kirk, J.A., Moore, J.R., Lehman, W., Campbell, S.G. (2025) Distinct mechanisms drive divergent phenotypes in hypertrophic and dilated cardiomyopathy associated TPM1 variants. J. Clin. Invest. Oct 22:e179135. doi: 10.1172/JCI179135. Online ahead of print. PMCID: PMC11645150.
10. Rynkiewicz, M.J., Childers, M.C., Karpicheva, O.E., Regnier, M, Geeves, M.A., Lehman, W. (2024) Myosin’s powerstroke transitions define atomic scale movement of cardiac thin filament tropomyosin. J. Gen. Physiol. 156: e202413538. doi: 10.1085/jgp.202413538. PMCID: PMC11010328.
11. Barry, M.E., Rynkiewicz, M.J., Pavadai, E., Viana A, Lehman W, Moore JR. (2024) Glutamate 139 of tropomyosin is critical for cardiac thin filament blocked-state stabilization. J. Mol. Cell Cardiol. S0022-2828(24)00004-X. doi: 10.1016/j.yjmcc.2024.01.004. PMCID: PMC11654406.
12. Lehman, W., Rynkiewicz M.J. (2023) Troponin-I-induced tropomyosin pivoting defines thin-filament function in relaxed and active muscle. J. Gen. Physiol. 155:e202313421. doi: 10.1085/jgp.202313387. PMCID: PMC10227645
13. Halder, S., Howland, H.L., Creso, J., Lehman, W.J., Moore, J.R., Rynkiewicz, M.J., Sewanan, L., Campbell, S.G. (2023) Mechanisms of pathogenicity in the hypertrophic cardiomyopathy doi: 10.1093/pnasnexus/pgad011. PMCID: PMC9991458
14. Doran, M.H., Rynkiewicz, M.J., Rasicci, D., Bodt, S.M.L, Barry, M., Bullitt, E., Yengo, C.M., Moore, J.R., Lehman, W. (2023) Conformational changes linked to ADP release from human cardiac myosin bound to actin-tropomyosin. J. Gen. Physiol. 155: e202213274. doi: 10.1085/jgp.202213267. PMCID: PMC9859928
15. Campbell, S.G., Moore, J.R., Rynkiewicz, M.J., Lehman, W. (2023) Multi-scale modeling will unravel connections between sarcomeric mutations and cardiomyopathies. J. Mol. Cell. Cardio. Plus. 2023 3: 100025. No PMCID provided
16. Doran, M.H., Rynkiewicz, M.J., Pavadai, E., Bodt, S.M.L, Rasicci, D., Moore, J.R., Yengo, C.M., Bullitt, E., Lehman, W. (2023) Myosin loop-4 is critical for optimal tropomyosin repositioning on actin during muscle activation and relaxation. J. Gen. Physiol. 155: e202213274. doi: org/10.1085/jgp.202213274. PMCID: PMC9723511
17. Rynkiewicz, M.J., Pavadai, E., Lehman, W. (2022) Modeling human cardiac thin filament structures. Front. Physiol. 13:932333. doi: 10.3389/fphys.2022.932333. PMCID: PMC9257132
18. Pavadai, E., Rynkiewicz, M.J., Yang, Z., Gould, I.R., Marston, S.B., Lehman, W. (2022) Modulation of cardiac thin filament structure by phosphorylated troponin-I analyzed by protein-protein docking and molecular dynamics simulation. Arch. Biochem. Biophys. 725:109282. doi: 10.1016/j.abb.2022.109282. PMCID: PMC10680062
19. Suphamungmee, W., Lehman, W., Morgan, K.M. (2022) Functional remodeling of the contractile smooth muscle cell cortex, a provocative concept, supported by direct visualization of cortical remodeling. Biology 11:662. doi: 10.3390/biology11050662. doi: 10.1016/j.xcrm.2021.100501.PMCID: PMC9138025
20. Teekakirikul, P., Zhu, W., Xu, X. …, Rynkiewicz, M.J., Lehman, W., … Lo, C.W. (2022) Genetic resiliency associated with dominant lethal TPM1 mutation causing atrial septal defect with high heritability. Cell Reports – Medicine 3:100501. doi: 10.1016/j.xcrm.2021.100501PMCID: PMC8861813
21. Doran MH, Lehman W. The Central Role of the F-Actin Surface in Myosin Force Generation. Biology (Basel). 2021 Nov 23;10(12):1221. doi: 10.3390/biology10121221. PMCID: PMC8698748
22. Lehman W, Pavadai E, Rynkiewicz MJ. C-terminal troponin-I residues trap tropomyosin in the muscle thin filament blocked-state. Biochem Biophys Res Commun. 2021 Apr 30;551:27-32. doi: 10.1016/j.bbrc.2021.03.010. PMCID: PMC8026701
23. Sewanan LR, Park J, Rynkiewicz MJ, Racca AW, Papoutsidakis N, Schwan J, Jacoby DL, Moore JR, Lehman W, Qyang Y, Campbell SG. Loss of crossbridge inhibition drives pathological cardiac hypertrophy in patients harboring the TPM1 E192K mutation J Gen Physiol. 2021 Sep 6;153(9):e202012640. doi: 10.1085/jgp.202012640. PMCID: PMC8321830
24. Racca AW, Rynkiewicz MJ, LaFave N, Ghosh A, Lehman W, Moore JR. M8R tropomyosin disrupts actin-binding and filament regulation. The beginning affects the middle and the end. J Biol Chem. 2020; 295(50):17128-17137. doi:10.1074/jbc.RA120.014713. PMCID: PMC7863880
25. Doran MH, Pavadai E, Rynkiewicz MJ, Walklate J, Bullitt E, Moore JR, Regnier M, Geeves MA, Lehman W. Cryo-EM and molecular docking shows myosin loop 4 contacts actin and tropomyosin on thin filaments. Biophys J. 2020; 119(4) 821-830. doi: 10.1016/j.bpj.2020.07.006 PMCID: PMC7451941
26. Pavadai E, Lehman W, Rynkiewicz MJ. Protein-protein docking reveals dynamic interactions of tropomyosin on actin filaments. Biophys J. 2020; 119(1):77-86. doi.org/10.1016/j.bpj.2020.05.017 PMCID: PMC7335911
27. Pavadai E, Rynkiewicz MJ, Ghosh A, Lehman W. Docking troponin T onto the tropomyosin overlapping domain of thin filaments. Biophys J. 2020;118(2):325‐336. doi:10.1016/j.bpj.2019.11.3393 PMCID: PMC6976810
28. Sundar S, Rynkiewicz MJ, Ghosh A, Lehman W, Moore JR. Cardiomyopathy mutation alters end-to-end junction of tropomyosin and reduces calcium sensitivity. Biophys J. 2020;118(2):303‐312. doi:10.1016/j.bpj.2019.11.3396 PMCID: PMC6976805
29. Viswanathan MC, Schmidt W, Franz P, Rynkiewicz MJ, Newhard CS, Madan A, Lehman W, Swank DM, Preller M, Cammarato A. A role for actin flexibility in thin filament-mediated contractile regulation and myopathy. Nat Commun. 2020;11(1):2417. doi:10.1038/s41467-020-15922-5 PMCID: PMC7229152
30. Lehman W, Rynkiewicz MJ, Moore JR. A new twist on tropomyosin binding to actin filaments: perspectives on thin filament function, assembly and biomechanics. J Muscle Res Cell Motil. 2020;41(1):23‐38. doi:10.1007/s10974-019-09501-5 PMCID: PMC6697252
31. Lehman W, Maéda Y. Introducing a special issue of the Journal of Muscle Research and Cell Motility on actin and actin-binding proteins. J Muscle Res Cell Motil. 2020;41(1):1‐2. doi:10.1007/s10974-019-09569-z, PMID: 31865487, PMCID: not provided by journal.
32. Lehman W, Rynkiewicz MJ, Moore JR. A new twist on tropomyosin binding to actin filaments: perspectives on thin filament function, assembly and biomechanics. J Muscle Res Cell Motil. 2020;41(1):23‐38. doi:10.1007/s10974-019-09501-5 PMCID: PMC6697252
Earlier publications are listed on: https://www.ncbi.nlm.nih.gov/myncbi/william.lehman.1/bibliography/public/
We collaborate with colleagues in several laboratories with similar interests including:
Dr. Aaron Beeler, Department of Chemistry, Boston University.
Dr. Lauren Brown, Department of Chemistry, Boston University.
Dr. Stuart Campbell, School of Engineering and Applied Science, Yale University.
Dr. Anthony Cammarato, Department of Medicine, Johns Hopkins University.
Dr. Roger Craig, Department of Radiology, University of Massachusetts Medical School.
Dr. Michael Geeves, School of BioSciences, University of Kent, Canterbury, UK.
Dr. Steven B. Marston, National Heart and Lung Institute, Imperial College, London, UK.
Dr. Jeffrey Moore, Department of Biological Sciences, University of Massachusetts-Lowell.
Dr. Raul Padron, Department of Radiology, University of Massachusetts Medical School.
Dr. Michael Regnier, Department of Bioengineering, University of Washington.
Links:
Contact Us
William J. Lehman
Department of Pharmacology, Physiology & Biophysics
Chobanian & Avedisian School of Medicine
700 Albany Street, W408E
Boston MA 02118-2526
Phone:(617) 358-8484
e-mail: wlehman@bu.edu