Glycosylation of influenza A virus hemagglutinin
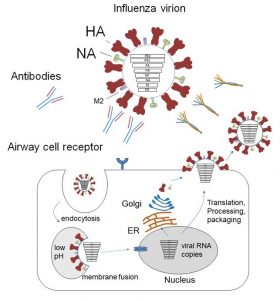
A glycoprotein may contain several sites of glycosylation, each of which is heterogeneous. As a consequence of glycoform diversity and signal suppression from non-glycosylated peptides that ionize more efficiently, typical reversed-phase LC-MS and bottom-up proteomics database searching workflows do not perform well for identification of site specific glycosylation for complex glycoproteins. We present an LC-MS system for enrichment, separation, and analysis of glycopeptides from complex glycoproteins (> four N-glycosylation sequons) in a single step. This system uses an on-line HILIC enrichment trap prior to reversed-phase C18-MS analysis. We demonstrated the effectiveness of the system using a set of glycoproteins including human transferrin (2 sequons), human alpha-1-acid glycoprotein (5 sequons), and influenza A virus hemagglutinin (9 sequons). The on-line enrichment renders glycopeptides the most abundant ions detected, thereby facilitating the generation of high quality data dependent tandem mass spectra. The tandem mass spectra exhibited product ions from both glycan and peptide backbone dissociation for a majority of glycopeptides tested using collisionally activated dissociation that served to confidently assign site specific glycosylation. We demonstated the value of our system to define site specific glycosylation using a hemagglutinin containing 9 N-glycosylation sequons from a single HILIC-C18-MS acquisition.
- Chang, D.; Hackett, W. E.; Zhong, L.; Wan, X. F.; Zaia, J. Measuring site-specific glycosylation similarity between influenza A virus variants with statistical certainty. Mol Cell Proteomics 2020, doi: 10.1074/mcp.RA120.002031. Pubmed Link
- Li, L.; Chang, D.; Han, L.; Zhang, X.; Zaia, J.; Wan, X. F. Multi-task learning sparse group lasso: a method for quantifying antigenicity of influenza A(H1N1) virus using mutations and variations in glycosylation of Hemagglutinin. BMC Bioinformatics 2020, 21, 182. Pubmed Link
- Chang, D.; Zaia, J. Why glycosylation matters in building a better flu vaccine. Mol Cell Proteomics 2019, 18, 2348-2358. Pubmed Link
- van Eijk, M.; Rynkiewicz, M. J.; Khatri, K.; Leymarie, N.; Zaia, J.; White, M. R.; Hartshorn, K. L.; Cafarella, T. R.; van Die, I.; Hessing, M.; Seaton, B. A.; Haagsman, H. P. Lectin-mediated binding and sialoglycans of porcine surfactant protein D synergistically neutralize influenza A virus. J Biol Chem 2018, 293, 10646-10662. Pubmed Link
- Khatri, K.; Klein, J. A.; White, M. R.; Grant, O. C.; Leymarie, N.; Woods, R. J.; Hartshorn, K. L.; Zaia, J. Integrated Omics and Computational Glycobiology Reveal Structural Basis for Influenza A Virus Glycan Microheterogeneity and Host Interactions. Mol Cell Proteomics 2016, 15, 1895-912. Pubmed Link
- Khatri, K.; Staples, G. O.; Leymarie, N.; Leon, D. R.; Turiák, L.; Huang, Y.; Yip, S.; Hu, H.; Heckendorf, C. F.; Zaia, J. Confident Assignment of Site-Specific Glycosylation in Complex Glycoproteins in a Single Step. J. Proteome Res. 2014, 13, 4347-4355. Pubmed Link
- Crouch, E.; Nikolaidis, N.; McCormack, F.; McDonald, B.; Allen, K.; Rynkiewicz, M.; Cafarella, T.; White, M.; Lewnard, K.; Leymarie, N.; Zaia, J.; Seaton, B.; Hartshorn, K. Mutagenesis of SP-D informed by evolution and xray crystallography enhances defenses against Influenza A Virus in vivo. J Biol Chem 2011, 286, 40681-92. Pubmed Link