Laboratory for Cortical Organization & Architectures
Our Research
We aim to identify and compare key morphological features of neurons, axons, and neuropil in diverse cortical areas, and to understand how these 1) enable area-specific functionality and 2) may be associated with differential vulnerability. Ongoing collaborations support investigations of white matter organization in macaque monkey (Dr. Alvaro Duque, Yale University) and investigation of pyramidal cell collaterals in prefrontal cortex of dorsolateral prefrontal cortex in normal and pathological brain aging (PI: Prof. Jennifer Luebke).
The general lab program is a continuation from decades of research on single axon topologies at the light microscopic level. A new direction, revisiting previous interests (e.g., Rockland and DeFelipe (2012)), is fine scale analysis of white matter organization in monkey and human. As a unique resource, we refer to several brains processed in uninterrupted serial sections after injections of the anterograde tracer biotinylated dextran amine (BDA). Click on the box below for more.
Underway are research conversations relating to neuroanatomy-tractography with several colleagues in the newly formed Society for International Tractography.
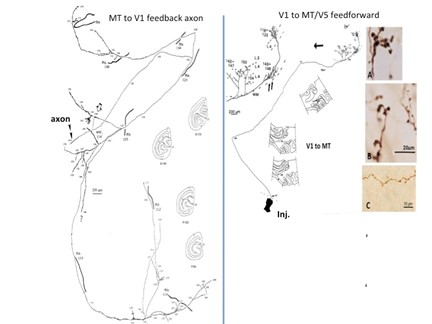
from Rockland and Knutson, 2000 / from Rockland, 1995
Using online databases for anatomically-based research.
Collaborator: Dr. Alvaro Duque (Yale University)

Student trainee: Silas Botham (Yale 2027, Neuroscience, B.S. & Education Studies Scholars Program)

Collaborator: Professor Martin Parent (Laval University; Quebec, Canada; Parent Lab
I. Background: Digital databases are increasingly important, both in the teaching and research domains. This is especially so for the nonhuman primate brain, where building a lab-based anatomical database is necessarily labor intensive and expensive. The availability of the NIH-funded MacBrain database (link above: Collection 6) is thus an important resource. I have been interacting with Dr. Alvaro Duque (PI) on ways to best position this for teaching and research purposes. Materials will be used for AN724 (Spring semester, co-instructor with Dr. Rushmore).
Completed: Neurochemical dissection of the Complex neurochemical microstructure of the stria terminalis in infant and adult macaque monkey. (Sakharkar, Rockland, and Duque, 2022)
As a prototype project, we have characterized the micro-structure of the stria terminalis (ST) in four infant and two adult macaque brains. The ST is a C-shaped fiber bundle, associated with the amygdala and bed nucleus of the stria terminalis (BNST).
As a teaching tool, this has offered opportunity for:
– learning to navigate the NHP brain in coronal tissue sections.
– identifying the ST in its characteristic location medial to the caudate nucleus (tail and body).
As a research tool, this has facilitated several specific results. For example:
– cellular populations, as visualized by NeuN.
– cellular subpopulations, as visualized by antibodies against neuropeptide Y, calretinin, calbindin, and others.
– compartmentalization, with reference to myelin-dense and myelin-sparse regions (visualized by antibodies against myelin basic protein).
– fiber orientation.
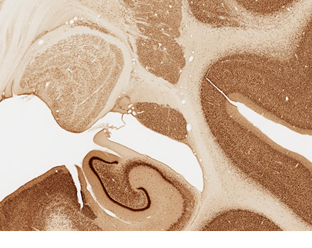
Below, location of the ST (ventral component), medial to the caudate nucleus. Coronal section reacted for NeuN, a pan-neuronal marker.
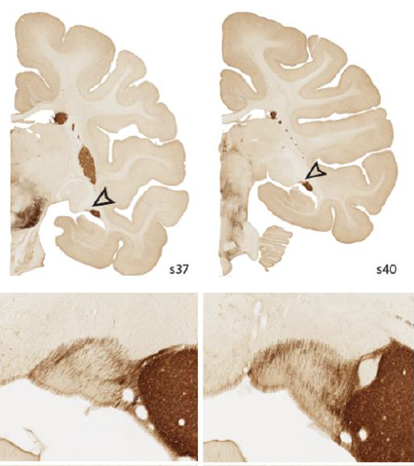
Two coronal sections, spaced 3.0mm apart, reacted for tyrosine hydroxylase (TH). ST is indicated by arrowheads. In the two higher magnification images (below), TH+ fibers are visualized. These travel along the circumference of the ST and concentrate in a beltlike pattern, cutting medial-lateral across the ST.
Back
Computational modeling of diverse cortical neurons and networks involved in working memory.
In collaboration with Christina Weaver (Franklin & Marshall College, Lancaster, PA) and Klaus Wimmer (Centre de Recerca Matemàtica, Barcelona, Spain), we build computational models that predict how alterations observed empirically with aging and neurodegeneration affect neuronal function. Our models operate at various scales, from individual pyramidal neurons, to local cortical networks, and across multiple brain areas, allowing us to understand the complex, nonlinear functions of the brain in new ways. The models are constrained by a wide range of our empirical data in rhesus monkeys and mice: electron and confocal microscopy, immunohistochemistry, electrophysiology, and behavioral testing.
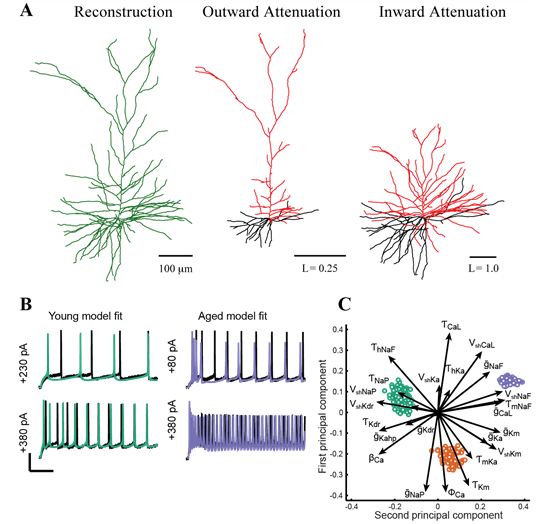
Computational modeling of pyramidal neurons. A: Morphoelectrotonic transforms demonstrate how signals attenuate outward from, or propagating in toward, the soma. Modified from Amatrudo et al (2012). B: Automated optimization enables fits of model parameters so that model outputs (green and blue lines) are similar to empirically measured in vitro voltage responses to current step injections. C: Analysis techniques including principal components analysis predict how individual ion channels may be affected by aging (green: young model population; red and blue, aged model populations). Panels B-C were modified from Rumbell et al. (2016).
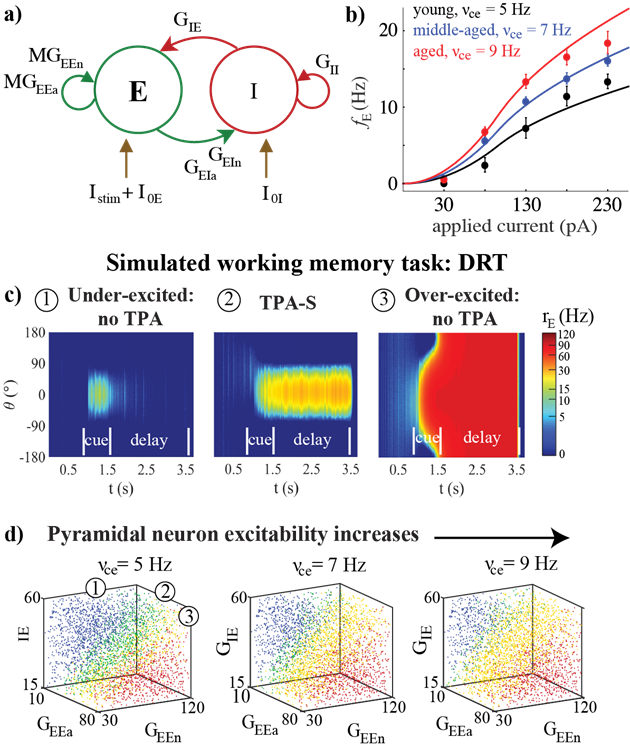
Effects of aging on a network model of the delayed response task (DRT). For details, see Ibañez et al. (2020).
Back



