Postdoctoral Position:
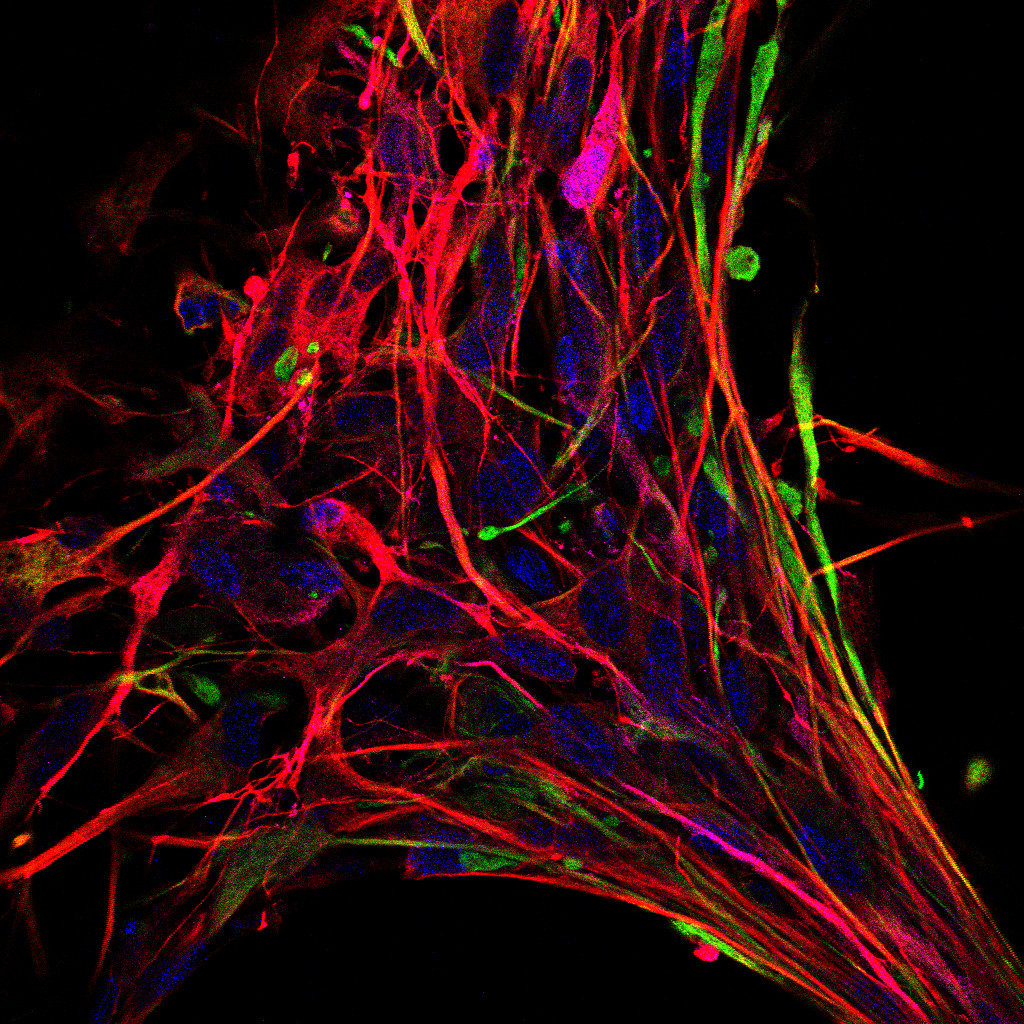
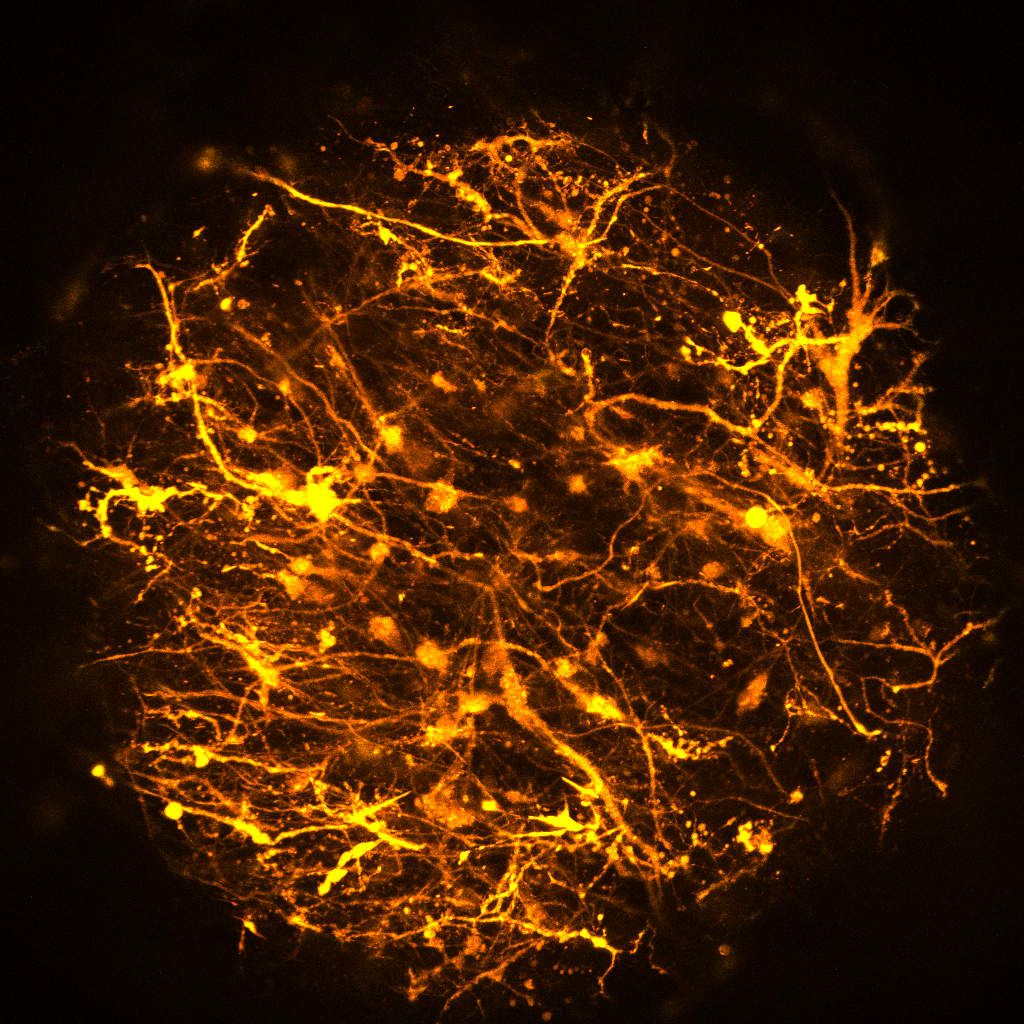
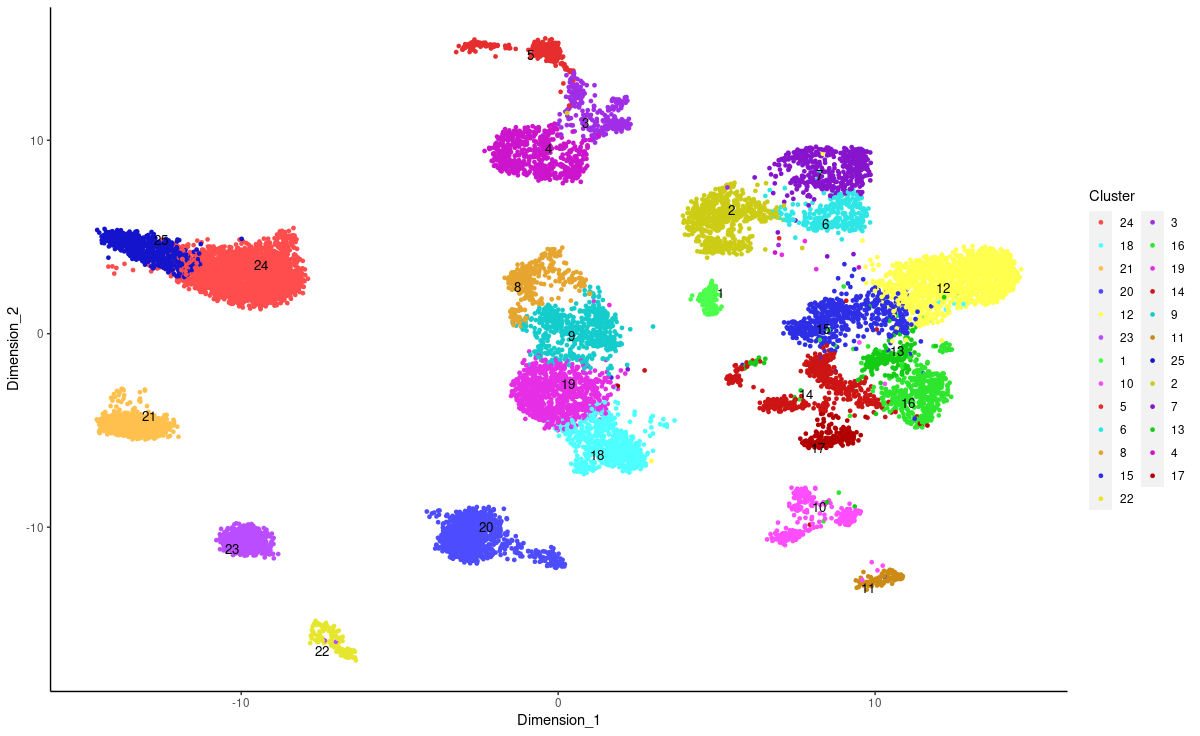
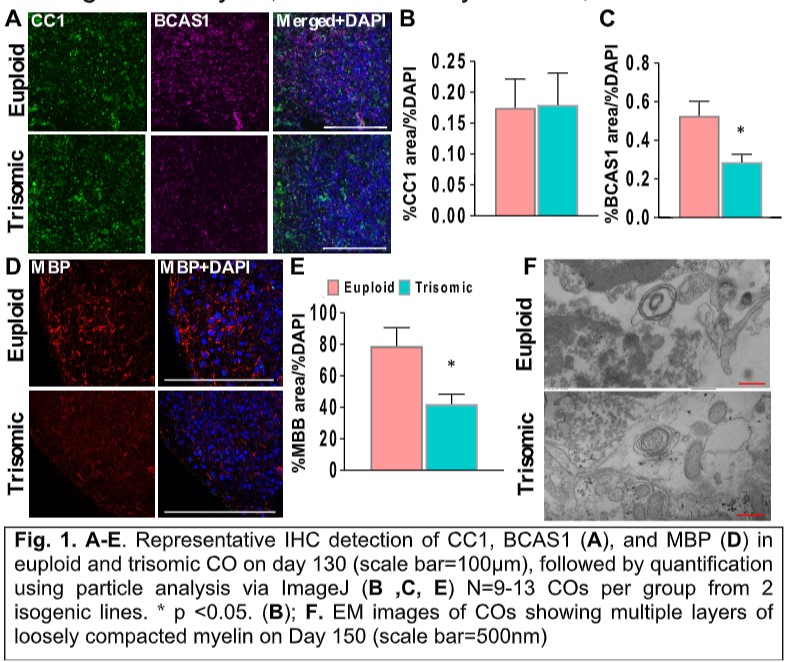
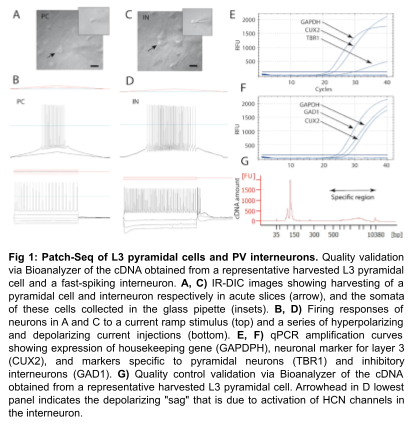
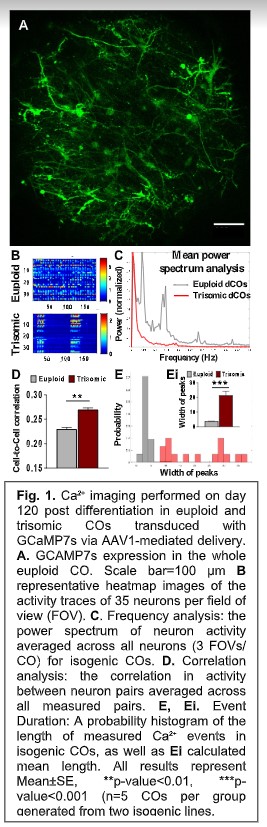
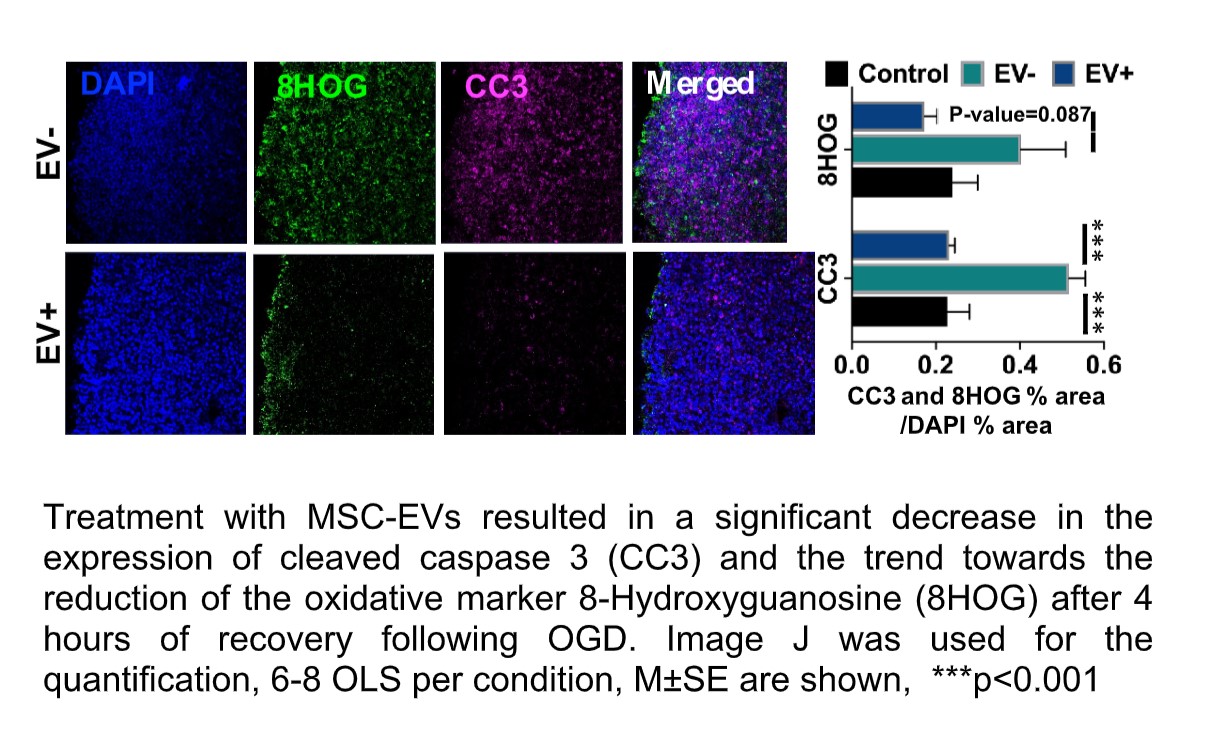
Job description: We are looking for an energetic and highly motivated molecular and cellular biologist or stem cell biologist to join our team. Research in the laboratory aims to understand the molecular and epigenetic mechanisms mediating brain cell abnormalities in Down Syndrome and its comorbidity with Alzheimer’s disease. Our studies are based on novel cutting-edge approaches utilizing human induced pluripotent stem cells (hiPSCs)-derived 3D in vitro models, including organoids and assembloids, CRISPR/Cas9 genome editing, as well as single-cell transcriptomic and multiome profiling. We are also in the process of establishing a platform for the assessment of the electrophysiological properties of cortical organoids for translational research.
The candidate will join a multidisciplinary and highly collaborative research team that includes computational geneticists, neuroscientists, and electrophysiologists in the department and within BU School of Medicine. The ideal candidate will be able to lead a project, take a part in the development of research proposals, and mentoring of graduate and undergraduate students. Bioinformatics experience is a plus.
In addition, those interested would have plentiful opportunities to teach graduate courses in neuroscience and anatomy.
The position is based on the recently-funded NIH grants and is available immediately.
As of March 6th, 2023